Our mission & value
OUR VISION
STERIMED’s business is organized around 3 main missions
INFECTION PREVENTION
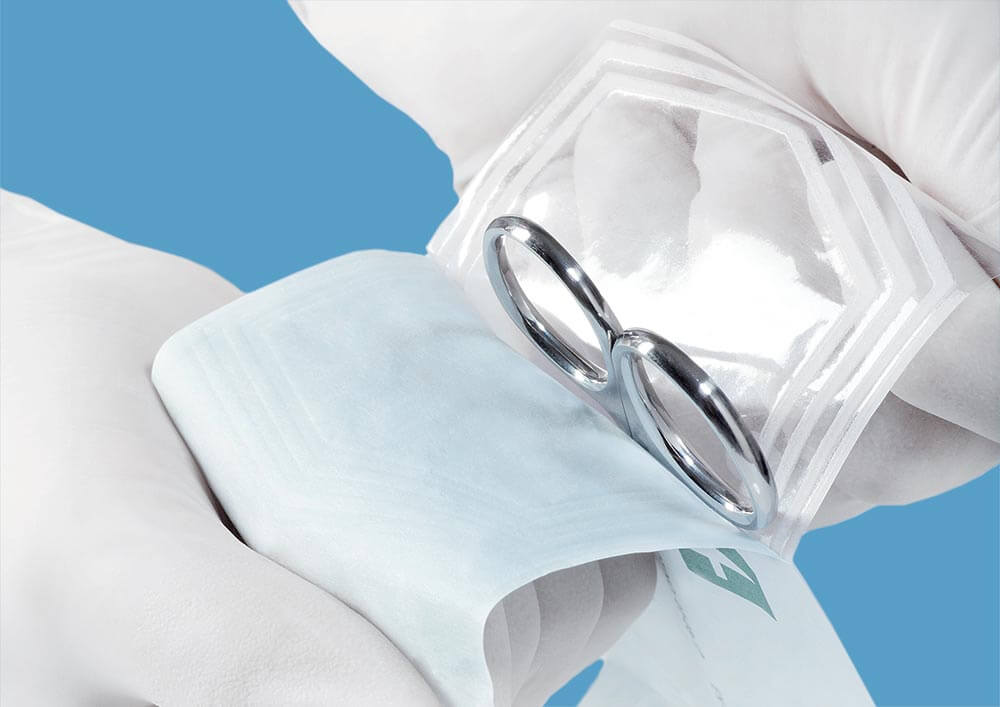
STERIMED products play a crucial role as sterile barrier to micro-organisms & physical protection of packed medical device. They must be safe to use for patients, hospital staff & people working on packaging lines. They must also be designed for aseptic opening at point of use and minimal impact on environement.
Supplied products are controlled for biocompatibility and bioburden.
Traceability is STERIMED other field of expertise, with software and harware solutions developed in partnership with hospital staff to help optimize workflow and patient safety in hospital premises.
STERIMED has developed a network of partnerships and expertise around the world which it relies on to ensure that it offers excellent solutions.

STERIMED products are recognized on the market for their first class safety and reliability.
PRODUCT INNOVATION, INDUSTRIAL PERFORMANCE & ENVIRONMENTAL FOCUS
Our products are manufactured with a high level of renewable ressource based content, and we use biomass energy in our medical paper product process.
We include eco-design strategies in product development phases and pay attention to consumption of raw materials, conditions of production in our industrial lines & product end life.
We are 100% healthcare focused group, in all our manufacturing sites.
Our R&D teams are experts in developing tailor made solutions to our consumer needs and innovative packaging materials wich serve as alternatives to non-renewable based products such as Tyvek®* or film-film packaging.
Our unique know how and global industrial set up makes us the partner of choice for companies looking at outsourcing industrial activities such as printing, slitting, sheeting or thermo-bonding of materials.
Awareness raising & education
As a leading actor in a market with constantly evolving standards, STERIMED is serious about making sure that standards and best practices are circulated to the various groups that the company works with on a daily basis:
- Personnel at sterilization centers
- Quality, production, and compliance divisions of medical device manufacturers
- Professional organizations representing actors in the packaging and conversion sector
- Pouch manufacturing partners
*Tyvek is registered trademark of E.I. du Pont de Nemours and Company

STERIMED is present on 5 continents and is a direct supplier to hospitals and medical device manufacturers around the world. The company has built up more than 40 years of knowledge of packaging standards and solutions, which enables us today to act as an advisor or trainer to our clients and end users of our products.

